All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know ALL.
The ALL Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the ALL Hub cannot guarantee the accuracy of translated content. The ALL Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The ALL Hub is an independent medical education platform, sponsored by Amgen and Autolus. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View ALL content recommended for you
AALL1331: blinatumomab improves outcomes for some low-risk pediatric and AYA patients with first relapse of B-ALL
Survival remains poor for first relapse of B-cell acute lymphoblastic leukemia (B-ALL) in children, adolescent, and young adult (AYA) patients, especially for early relapses and as such there is a need to improve current treatment approaches for these patients. Standard treatment for early relapse includes 1 month of reinduction chemotherapy, followed by consolidation with two additional cycles of intensive chemotherapy before stem cell transplantation, with the goal to achieve minimal residual disease (MRD) negativity prior to the transplant. For late relapses, high MRD post-reinduction has been associated with dismal prognosis, hence consolidation is the same as for early relapses, whereas patients with low MRD levels can often be cured with consolidation and maintenance chemotherapy.
Blinatumomab is a bispecific T-cell engaging antibody that targets CD19 and is FDA approved for the treatment of children and adults with relapsed/refractory B-ALL. The FDA has also granted accelerated approval to blinatumomab for MRD positive B-ALL.
The objective of the randomized phase III AALL1331 study by the Children’s Oncology Group was to determine if substituting blinatumomab for intensive consolidation chemotherapy improves survival in first relapse of childhood/AYA B-ALL. Results from the low-risk cohort of the AALL1331 study were presented by lead investigator Patrick A. Brown at the 63rd American Society of Hematology (ASH) Annual Meeting and Exposition in December 2021 as part of the “Acute lymphoblastic leukemias: therapies, excluding transplantation and cellular immunotherapies” oral abstract session.1
Study design
The study was conducted in 170 Children’s Oncology Group centers. In total, 255 low-risk patients were randomized from January 2015 to September 2019 (Figure 1). Any first relapse B-ALL patients aged 1–30 years regardless of remission duration were eligible to take part in the trial. Exclusion criteria included Down’s syndrome, Philadelphia chromosome positive ALL, prior transplant, and prior treatment with blinatumomab. The primary and secondary endpoints were intent-to-treat 4-year disease-free survival (DFS) from the time of randomization and overall survival (OS), respectively.
- All patients received the same reinduction regimen based on Block 1 of UKALLR3 mitoxantrone arm.3
- After reinduction patients were evaluated and risk assigned as treatment failure for ≥25% marrow blasts and/or non-clearance of extramedullary (EM) disease, high risk for early relapses and low risk for late relapses.
- Bone marrow (BM) MRD levels after reinduction were used to further stratify late relapses into low risk with MRD <0.1% and intermediate risk with MRD ≥0.1%.
- This article will focus on the previously unpublished results of the post reinduction randomized treatment of the low-risk group.
- To ensure a balance of potentially confounding factors randomization was stratified according to the site of relapse BM versus isolated EM (iEM) and for BM relapses by the level of MRD.
- Patients with central nervous system (CNS) disease at study entry either isolated or combined with BM relapse received 1,800 cGy of cranial radiation after the first maintenance cycle.
- Patients with testicular leukemia at relapse that persisted after reinduction received 2,400 cGy testicular radiation during the second block of chemotherapy.
Figure 1. Study design*
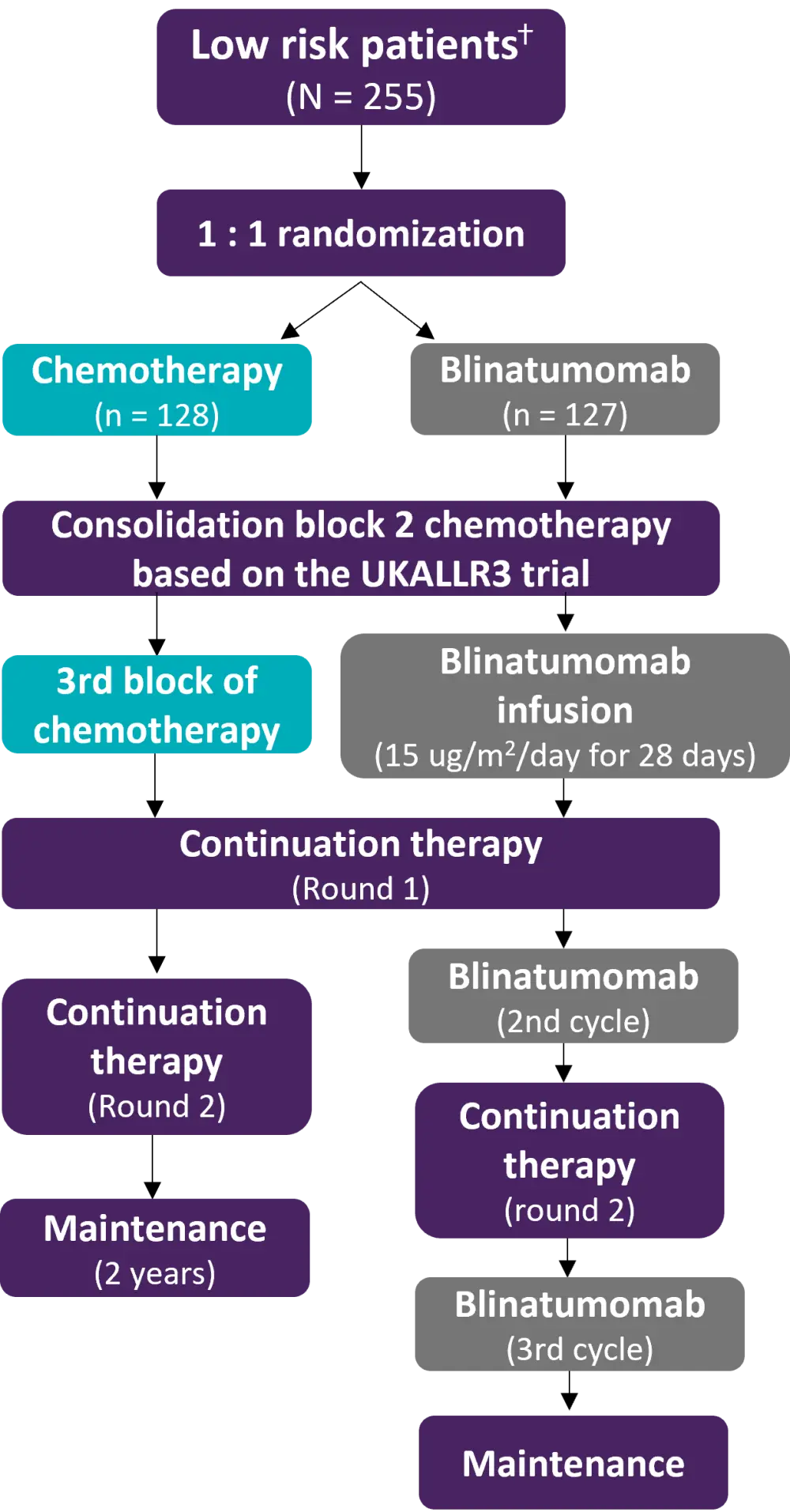
* Adapted from Brown.1
ⴕ Low risk defined as BM±EM relapse ≥36 months or iEM relapse ≥18 months from initial diagnosis, and low (<0.1%) BM MRD at the end of reinduction chemotherapy.
Results
Patient characteristics are summarized in Table 1. The two arms were well balanced for clinically significant baseline characteristics.
Table 1. Baseline patient characteristics*
|
BM, blood marrow; CNS, central nervous system; EM, extramedullary; IEM, isolated extramedullary; MRD, minimal residual disease. |
|||
|
|
Blinatumomab arm |
Chemotherapy arm |
Total (n = 255) |
|---|---|---|---|
|
Site of relapse, % |
|||
|
BM ± EM |
87 |
87 |
174 |
|
BM |
70 |
72 |
142 |
|
BM + CNS |
10 |
10 |
20 |
|
BM + testes |
7 |
5 |
12 |
|
IEM |
40 |
41 |
81 |
|
CNS |
30 |
33 |
63 |
|
CNS + testes |
1 |
0 |
1 |
|
Testes |
9 |
8 |
17 |
|
End of Block 1 of chemotherapy MRD, % |
|||
|
<0.01% |
108 |
109 |
217 |
|
≥0.01% and <0.1% |
19 |
19 |
38 |
|
Age at relapse, % |
|||
|
<12 years |
71 |
75 |
146 |
|
12 to 17 years |
38 |
37 |
75 |
|
≥18 years |
18 |
16 |
34 |
Adverse events
When comparing the rates of clinically significant adverse events, patients in the standard chemotherapy arm had strikingly higher rates of Grade III–IV febrile neutropenia, infection, sepsis, anemia, and mucositis relative to blinatumomab (Figure 2).
Figure 2. Comparison of the incidence of AEs in low-risk patients treated with 3rd block chemotherapy versus 1st cycle of blinatumomab*
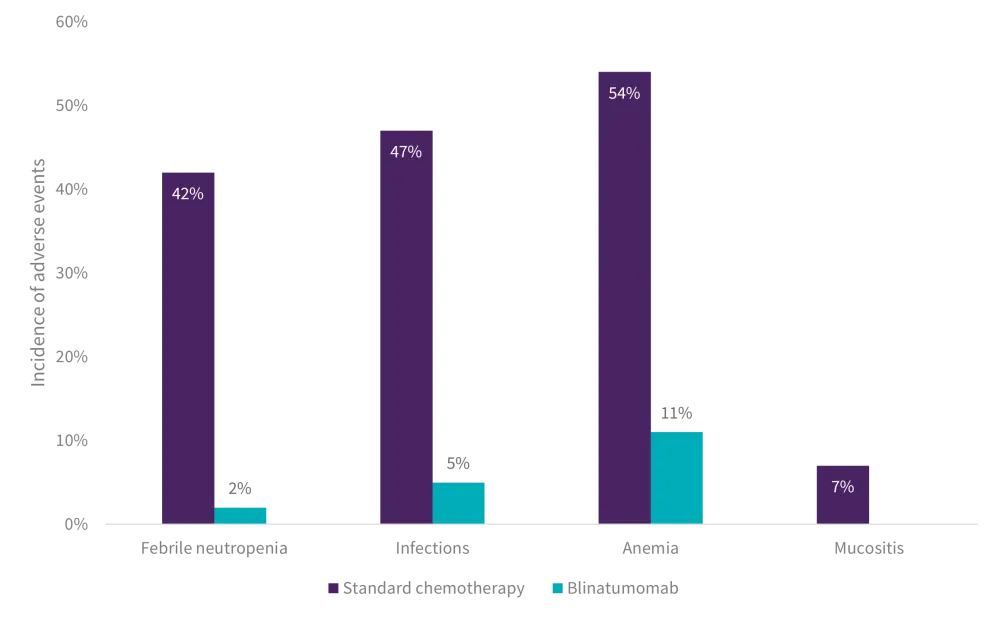
AE, adverse event.
*Adapted from Brown.1
Blinatumomab was well tolerated. Cytokine release syndrome, seizures, and other neurotoxicities (e.g., cognitive disturbance, tremor, ataxia, and dysarthria) were generally low-grade and fully reversible, with the highest rates in Cycle 1 and lower rates in subsequent cycles (Table 2).
Table 2. Blinatumomab-related AEs
|
AE, adverse event; CRS, cytokine release syndrome. |
|||
|
AE, % |
Cycle 1 |
Cycle 2 |
Cycle 3 |
|---|---|---|---|
|
CRS |
12 |
7 |
7 |
|
Seizure |
3 |
1 |
1 |
|
Other neurotoxicity |
19 |
9 |
5 |
Efficacy analysis
- Overall, 88% of observed DFS events were second relapses, 11% were deaths in complete remission, and one was attributed to a secondary malignant neoplasm.
- While there were fewer events in the blinatumomab arm (standard chemotherapy arm: 47; blinatumomab arm: 37) the difference in DFS did not meet the prespecified efficacy Z value.
- With a median follow-up of 2.9 years, 4-year DFS was 48.2 ± 6.0% for standard chemotherapy vs. 61.2 ± 5.5% for blinatumomab (p = 0.154).
- The 4-year OS did not significantly differ between the arms (91.6 ± 3.0% for blinatumomab vs 83.3 ± 4.5% for standard chemotherapy; p = 0.096).
Stratified analysis
Striking differences in DFS and blinatumomab efficacy were noted according to site of first relapse. Table 3 compares the difference between the efficacy of blinatumomab for BM ± EM vs iEM relapse.
Table 3. DFS events by the site of first relapse*
|
BM, blood marrow; DFS, disease-free survival; EM, extramedullary; IEM, isolated extramedullary; SMN, second malignant neoplasm. *Data from Brown.1 |
||||
|
|
BM ± EM |
IEM |
||
|---|---|---|---|---|
|
DFS events, n |
Standard |
Blinatumomab |
Standard |
Blinatumomab |
|
No events |
62 |
73 |
19 |
17 |
|
Secondary |
22 |
12 |
19 |
21 |
|
SMN |
0 |
0 |
0 |
1 |
|
Deaths |
3 |
2 |
3 |
1 |
- Second relapse rates were markedly higher for iEM relapses compared to BM relapses (46% and 43% for iEM relapse vs 25% and 14% for BM ± EM relapse in the standard chemotherapy and blinatumomab arms, respectively).
- A reduction in relapse for blinatumomab relative to standard chemotherapy was seen only for BM ± EM relapses.
- For BM ± EM relapses, 4-year DFS was 74.0 ± 6.4% for blinatumomab vs 51.8 ± 7.9% for standard chemotherapy (p = 0.016), and 4-year OS was 96.6 ± 2.5% for blinatumomab vs 84.4 ± 5.6% for standard chemotherapy (p = 0.013).
- For iEM relapses, 4-year DFS was 34.2 ± 8.6% for blinatumomab vs 39.3 ± 8.5% for standard chemotherapy (p = 0.73), and 4-year OS was 81.7 ± 7.0% for blinatumomab vs 80.8 ± 7.2% for standard chemotherapy (p = 0.61).
- The difference in DFS between BM ± EM and iEM patients was driven by excess of second relapse in isolated CNS relapse patients, with no difference by treatment arm (Figure 3).
Figure 3. Second relapse rate by site of first relapse*
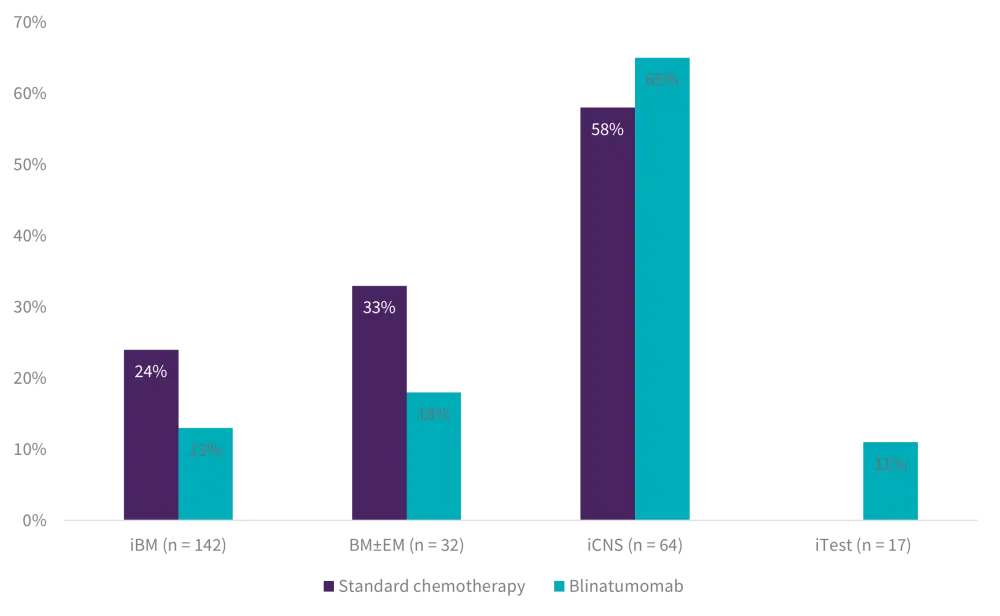
BM, bone marrow; EM, extramedullary; iBM, isolated bone marrow; iCNS, isolated central nervous system; iTest, isolated testes.
*Adapted from Brown.1
- Significant predictors of improved DFS in Cox multivariable regression for BM ± EM relapses included randomization to blinatumomab (hazard ratio [HR], 0.54; 95% confidence interval [CI], 0.27–1.1; p = 0.034), ≥18 years at enrollment (HR, 3.9; 95% CI, 1.6–9.5; p = 0.008), and relapsing ≥4 years from original diagnosis (HR, 0.43; 95% CI, 0.20–0.91; p = 0.027).
- For isolated CNS relapse the only significant predictor of improved DFS in Cox multivariable regression was relapsing ≥3 years from original diagnosis (HR, 0.42; 95% CI, 0.19–0.99; p = 0.016).
- The only predictor of DFS in IEM patients was site of first relapse (HR for testes vs CNS, 0.19; 95% CI, 0.04–0.87; p = 0.015).
Conclusion
The study demonstrated that for children and AYA with low risk first relapse of B-ALL there was no significant difference in outcome for the entire population. Notably, the blinatumomab arm was superior to the standard chemotherapy arm for patients with BM with or without combined EM relapse, establishing this regimen as a new standard therapy for this patient subset. However, in this trial blinatumomab was not superior for iEM relapse. Furthermore, isolated CNS relapse patients had a significantly high relapse rate on both arms proving that better treatments are needed for these patients.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

