All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know ALL.
The ALL Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the ALL Hub cannot guarantee the accuracy of translated content. The ALL Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The ALL Hub is an independent medical education platform, sponsored by Amgen and Autolus. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View ALL content recommended for you
ASH 2023: Optimizing conditioning regimens for allo-HSCT in ALL
Question 1 / 1
In the study investigating CD7 chimeric antigen receptor T-cell therapy as bridging to allogeneic hematopoietic stem cell transplantation in relapsed/refractory T-cell acute lymphoblastic leukemia, which of the following factors was associated with a worse overall and disease-free survival?
A
Total body irradiation-based conditioning
B
CD7 CAR T-cell therapy
C
TP53
D
Chronic graft-versus-host disease
In this article, we summarize three key trials focused on optimizing conditioning regimens for allogeneic hematopoietic stem cell transplantation (allo-HSCT) for patients with acute lymphoblastic leukemia (ALL), presented by Kalwak.1, Ji.2, Li.3 at the 65th American Society of Hematology (ASH) Annual Meeting and Exposition.
Busulfan- vs treosulfan-based conditioning regimen in children with ALL undergoing allo-HSCT1
Results from the prospective international FORUM trial comparing busulfan versus treosulfan-based conditioning regimens in patients aged ≤18 years with ALL undergoing allo-HSCT across both randomizing and non-randomizing countries between 2013−2018 were presented by Kalwak.1
Results
A total of 308 patients were included in the study, 128 of whom received treosulfan/thiotepa/fludarabine and 180 received busulfan/thiotepa/fludarabine. Baseline characteristics were balanced between the two cohorts.
Outcomes
At a median follow-up of 4.2 years, there was no difference in the overall survival (OS), event-free survival (EFS), cumulative incidence of relapse (CIR), and non-relapse mortality (NRM) between the two groups, with similar 5-year rates (OS: 69% in both; EFS: 57% and 55% in busulfan vs treosulfan; CIR: 34% and 36% in busulfan vs treosulfan; and NRM of 9% in both groups). There was one case of secondary malignancy seen in the treosulfan cohort and one case of fatal liver venoocclusive disease in the busulfan group.
Moreover, there were no statistical differences in the cumulative incidence of both acute graft-versus-host disease (aGvHD) and chronic GvHD (cGvHD) between the two groups, though there was a trend towards a higher incidence of cGvHD in the busulfan group. Additionally, the GvHD-free survival rate was similar between the two cohorts.
For patients in complete remission (CR) 1 and CR2, there were no differences in the OS, EFS, CIR, NRM, GvHD-free relapse-free survival , and cGVHD incidence between the two conditioning regimens; however, the 5-year EFS rates were lower (75% and 66% in busulfan and treosulfan group, respectively at CR1 vs 42 and 40%, respectively at CR2) and the 5-year CIR was higher (17% and 27% in busulfan and treosulfan group, respectively at CR1 vs 48% and 47%, respectively at CR2) for those in CR2 compared with CR1. Overall, there were no significant differences in outcomes by country, but a lower OS and EFS was observed in non-randomized vs randomized countries.
Presenter’s conclusion
This prospective study reported comparable long-term outcomes between busulfan and treosulfan-based conditioning regimens in children with ALL undergoing allo-HSCT. As such, both regimens can be safely and effectively used worldwide for patients aged >4 years, located in centers or countries unable to deliver total body irradiation regimen.
Short-term blinatumomab as bridging therapy for allo-HSCT in R/R B-ALL2
Results from a single-arm, prospective study of short-term blinatumomab as bridging therapy to allo-HSCT in patients with B-ALL and low leukemia burden was presented by Ji.2 Patients received a total dose 175 μg dose of blinatumomab followed by myeloablative conditioning with cytarabine, fludarabine, busulfan, and chidamide. All patients received peripheral stem cell grafts and posttransplant cyclophosphamide. The study endpoints included survival and disease relapse rates.
Results
A total of 23 patients were included in the study, 50% of whom had Philadelphia chromosome-positive ALL, and over 30% with a 1% leukemia burden prior to therapy.
Response and toxicities
All patients achieved CR and measurable residual disease clearance by flow cytometry; however, 5 out of the 11 patients with Philadelphia chromosome-positive ALL did not achieve measurable residual disease clearance by real-time quantitative polymerase chain reaction. The median interval between blinatumomab and allo-HSCT was 5.5 days. All patients were fully engrafted by Day 30 with a median neutrophil and platelet recovery of 12 and 13 days, respectively.
Grade 1 cytokine release syndrome occurred in 39.1%, with no Grade 2 or higher cases. There was one case of Grade 1 immune effector cell-associated neurotoxicity syndrome and Grade 2 tremors.
Relapse and survival
There was one case of relapse reported at 24.3 months, and one death at 7.2 months due to COVID-19 associated with pulmonary aspergillosis. At a median follow-up of 10.2 months, the 1-year relapse-free survival and OS was both 94.4%. The cumulative incidence of Grade 2–4 or Grade 3–4 aGvHD was 17.4% and 8.7%, respectively. The 1-year cumulative incidence of any grade cGvHD was 27.8%, with a 1-year cumulative incidence of moderate cGvHD in 7.7% and no cases of severe cGvHD. The 1-year cumulative incidence of cytomegalovirus reactivation was 47.8%, and the 1-year Grade 2 or higher hemorrhagic cystitis was seen in 26.1%.
Presenter’s conclusion
This study showed that the short-term use of blinatumomab as bridging therapy to allo-HSCT is safe and effective in patients with B-ALL and low leukemia burden, with an acceptable relapse rate and no unexpected adverse events reported. A large-scale, multicenter, prospective study is needed to confirm the long-term effectiveness of this approach.
CD7 CAR T-cell therapy as bridging to allo-HSCT in R/R T-ALL3
The long-term outcomes of CD7 chimeric antigen receptor (CAR) T-cell therapy as bridging to allo-HSCT in patients with R/R T-cell ALL was presented by Li.3 The risk factors for prognosis after allo-HSCT was analyzed.
Results
A total of 90 patients were included in the study who were subdivided into three groups: 32 who were sensitive to chemotherapy and achieved CR before transplant (Group A); 41 who were resistant to chemotherapy, in non-remission pre-HSCT, and received CD7 CAR T-cell therapy before allo-HSCT (Group B), and 17 patients were in non-response before allo-HSCT (Group C).
Relapse and survival
All patients achieved durable engraftment. There was no significant difference in the incidence of aGvHD, cGvHD, and infections between the three groups.
At a median follow-up of 25 months, the 2-year OS and disease-free survival (DFS) in Group B was similar to Group A but much higher than Group C. The 2-year CIR in Group B was similar to Group A but much lower when compared with Group C. There was no significant difference in treatment-related mortality among the three groups (Figure 1).
Figure 1. 2-year OS, DFS, relapse and TRM in Group, A, B, and C*
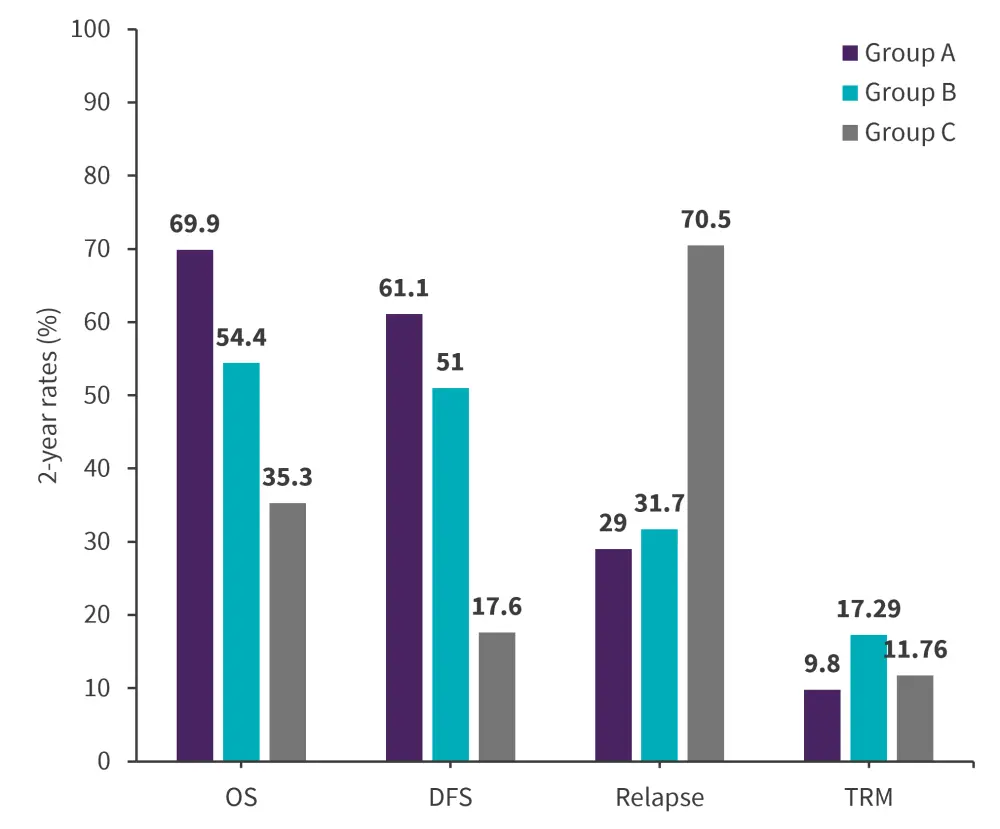
DFS, disease-free survival; OS, overall survival; TRM, treatment-related mortality.
*Adapted from Li.3
The most common germline mutations across the three groups were EP300, ATM, F7, KIT, and NCF2; these did not significantly impact the prognosis post allo-HSCT. The most common somatic mutations were NOTCH1, JAK3, TP53, JAK1, and NRAS; TP53 and MED12 mutations were associated with a significantly higher risk of relapse and death.
Multivariate analyses showed that CD7 CAR T-cell therapy, total body irradiation (TBI)-based conditioning, and cGvHD were independent positive prognostic factors for OS. Conversely, aGvHD and TP53 were independent negative prognostic factors for OS. Moreover, CD7 CAR T-cell therapy and TBI-based conditioning was positively associated with DFS, but TP53 was negatively associated with DFS.
Presenter’s conclusion
This study demonstrated that CD7 CAR T-cell therapy as bridging therapy to allo-HSCT can significantly improve DFS outcomes for patients with chemo-resistant T-cell ALL, as well as achieve comparable outcomes to patients with chemotherapy-sensitive disease posttransplant. The study highlighted the positive impact of factors such as CD7 CAR T-cell therapy and TBI-based conditioning on survival, whereas TP53 mutations are associated with an unfavorable prognosis.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

