All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know ALL.
The ALL Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the ALL Hub cannot guarantee the accuracy of translated content. The ALL Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The ALL Hub is an independent medical education platform, sponsored by Amgen and Autolus. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View ALL content recommended for you
Reduced chemotherapy and chemotherapy-free regimens for patients with ALL
Do you know... Which of the following statements about reduced chemotherapy and chemotherapy-free regimens for the treatment of ALL is false?
Limitations of chemotherapy in the treatment of ALL
Traditionally, chemotherapy has played an integral role in the treatment of acute lymphoblastic leukemia (ALL) and is key in induction, consolidation, and maintenance regimens.1,2 However, it has a number of limitations. As it is a non-selective cytotoxic therapy, it does not specifically act on cancer cells, and a number of adverse events (AEs) can result from its effect on healthy cells, such as nephrotoxicity, hepatotoxicity, neurotoxicity, cardiotoxicity, and hematologic toxicities.3 This off-target DNA damage in healthy tissues can also lead to accelerated cellular aging.3
Older patients are generally less able to tolerate chemotherapy than younger patients, likely due to reduced drug clearance and smaller volumes of drug distribution.3 These differences may result from poorer liver and kidney function, and lower total body water percentages.3 The prolonged organ toxicity, immunosuppression, and amplification of chronic diseases that accompany chemotherapy impact quality of life, particularly in elderly patients who are less able to care for themselves.4
Following chemotherapy treatment, remaining cancer cells (measurable residual disease [MRD]) adopt slow cycling, causing reduced susceptibility to conventional chemotherapy and subsequent disease persistence.5 Achievement of MRD-negativity is associated with better survival outcomes in patients with cancer, supporting investigation into therapeutics that either target MRD cells or prevent MRD occurrence.5 Given these limitations, there has been a shift towards investigating strategies that would reduce reliance on chemotherapy-based treatment options to improve patient tolerance, reduce systemic AEs, and reduce risk of MRD cycling.1,5,6
Immune-based therapeutic strategies in reduced chemotherapy and chemotherapy-free ALL regimens
Immunotherapies, such as bispecific T-cell engagers (BiTEs), antibody–drug conjugates (ADCs), and chimeric antigen receptor (CAR) T-cell therapies, utilize the body’s own immune system to recognize and attack cancer cells.6 A number of immunotherapies are currently indicated as monotherapies for the treatment of relapsed/refractory (R/R) ALL (Table 1). More recently, immunotherapies are being investigated in combination with chemotherapy or tyrosine kinase inhibitors (TKIs) as first-line treatment for ALL.1
Table 1. Immunotherapies approved as monotherapy for the treatment of ALL
| Approved indication | ||
| Drug | FDA | EMA |
| Blinatumomab7,8 | Adults and children aged ≥1 month with:
| Adults with:
Children aged ≥1 month with:
|
| InO9,10 | Adults and children aged ≥1 year with:
| Adults with:
|
| Brexu-cel11,12 | Adults with:
| Adults aged ≥26 years with:
|
| Tisagenlecleucel13,14 | Patients aged ≤25 years with:
| Patients aged ≤25 years with:
|
| Obe-cel15,16 | Adults with:
| Adults aged ≥26 years with:
|
ALL, acute lymphoblastic leukemia; allo-HSCT, allogeneic hematopoietic stem cell transplantation; B-ALL, B-cell ALL; BCP, B-cell precursor; brexu-cel, brexucabtagene autoleucel; EMA, European Medicines Agency; FDA, U.S. Food and Drug Administration; InO, inotuzumab ozogamicin; MRD, measurable residual disease; ND, newly diagnosed; obe-cel, obecabtagene autoleucel; Ph, Philadelphia chromosome; R/R, relapsed/refractory; TKI, tyrosine kinase inhibitor.
BiTEs
BiTEs are comprised of two arms that bind to T cells and tumor cells, triggering T-cell activation and subsequent selective cytotoxicity (Figure 1).17
Figure 1. Mechanism of action of a BiTE*
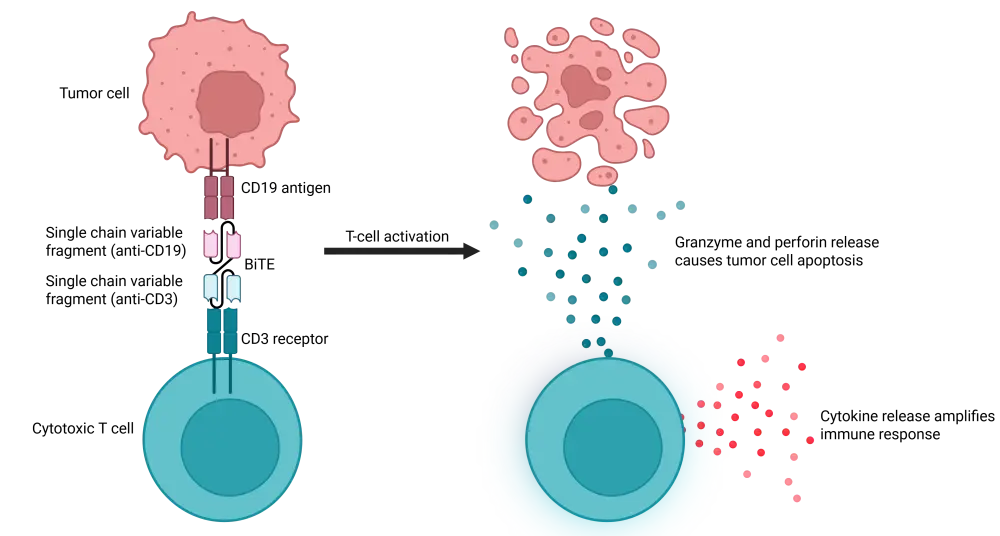
Blinatumomab is approved as a monotherapy by the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) for the treatment of specific ALL subtypes (Table 1), and has become a key therapeutic option for patients with R/R disease following positive findings from the MT103-211 (NCT01466179), TOWER (NCT02013167), and ALCANTARA (NCT02000427) trials.7,8,19 It is recommended over chemotherapy for the treatment of adolescent and young adult patients with R/R B-cell ALL (B-ALL) by the American Society of Hematology (ASH).20 The use of blinatumomab in combination with chemotherapy or other targeted therapies is currently being assessed in clinical trials, largely focusing on consolidation or maintenance therapy; however, more recently, evidence has suggested that blinatumomab may be effective when integrated into first-line regimens for newly diagnosed (ND) ALL.1
In the ongoing phase III ECOG-ACRIN-E1910 trial (NCT02003222) in patients with ND B-ALL, at a median follow-up of 43 months, an overall survival (OS) benefit was demonstrated at 3 years in patients who received blinatumomab + chemotherapy vs chemotherapy alone (85% vs 68%; p = 0.002).21 Grade 3 non-hematologic AEs occurred at similar rates between treatment arms (43% vs 36%); however, Grade ≥3 treatment-related neurologic events were more common with blinatumomab + chemotherapy vs chemotherapy alone (23% vs 5%; p < 0.001).21
The combination of blinatumomab + chemotherapy is also being investigated in the ongoing phase III AALL1731 trial (NCT03914625) in adults and in pediatric patients aged ≥1 and <10 years with B-ALL. Results from the pediatric cohort of this study showed that 3-year disease-free survival (DFS) was greater in patients who received blinatumomab + chemotherapy vs chemotherapy alone (96.0% vs 87.9%; p = 0.00004).22 During blinatumomab cycles, the incidence of Grade ≥3 blinatumomab-associated toxicities, such as cytokine release syndrome (CRS; 0.2%), sepsis and catheter-related infections (2.9%), or seizures (0.8%), was low.22
The phase II GIMEMA LAL2317 trial (NCT03367299), evaluating the impact of sequential chemotherapy and blinatumomab on MRD response and survival in patients with ND Philadelphia chromosome-negative (Ph−) B-cell precursor ALL (BCP-ALL), showed that 93% of patients achieved MRD negativity after the first cycle of blinatumomab.23 Stratification by age revealed improved complete response (CR; 94%, 92%, and 64% for 18–40, 40–55, and >55 year olds, respectively; p < 0.001) and OS (76%, 74%, and 49%, respectively; p = 0.00013) in younger vs older patients.23
Across the ECOG-ACRIN-E1910, AALL1731, and GIMEMA LAL2317 trials, blinatumomab demonstrated efficacy and tolerability in adult and pediatric patients with ND B-ALL and BCP-ALL, supporting its use outside of the R/R setting.
ADCs
ADCs are synthetic molecules that target antigens expressed exclusively on tumor cells or are overexpressed during carcinogenesis (Figure 2).24 Inotuzumab ozogamicin (InO) is an FDA- and EMA-approved humanized immunoglobulin G4 anti-CD22 antibody that is indicated for the treatment of adult and pediatric patients with R/R BCP-ALL (Table 1).9,10 Research into InO has mostly been in the R/R setting, but more recent studies have expanded into first-line combinations with chemotherapy and/or other immunotherapies.25
Figure 2. Mechanism of action of an ADC*
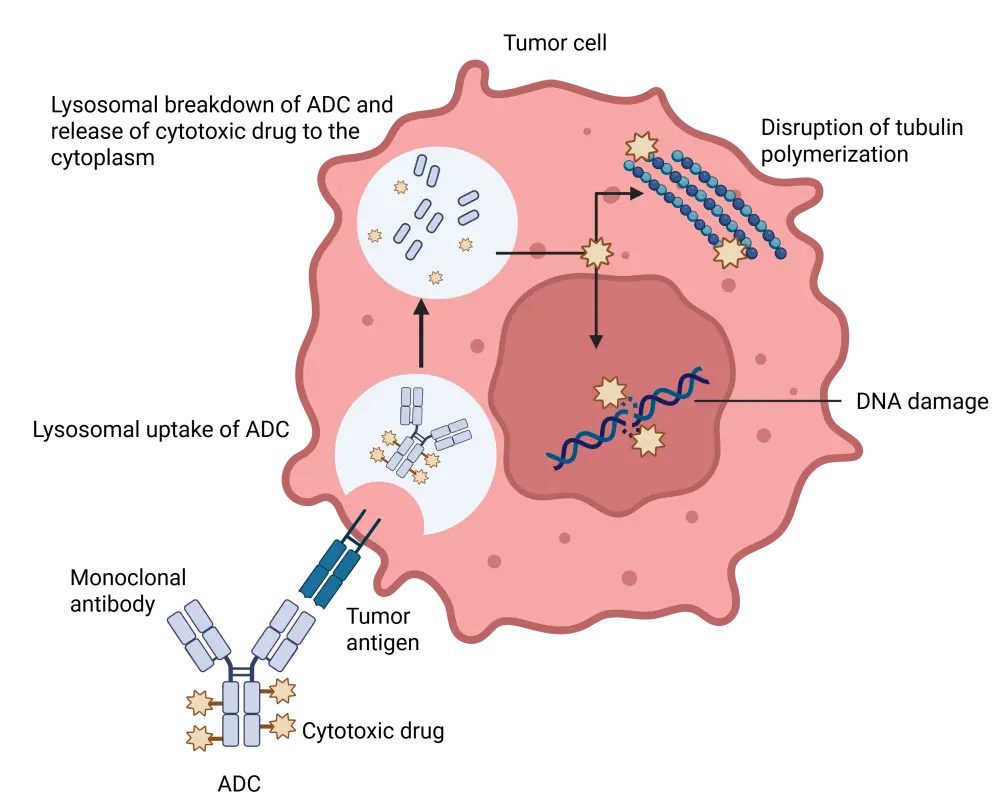
In the phase III INO-VATE study (NCT01564784), comparison of InO with chemotherapy in adult patients with R/R CD22+ B-ALL found higher CR rates (80.7% vs 29.4%; p < 0.001), MRD negativity rates (78.4% vs 28.1%; p < 0.001), and median OS (7.7 months vs 6.7 months; p = 0.04).26 Grade ≥3 AEs occurred in 46% of patients receiving InO and 43% of patients receiving chemotherapy.26 Hepatic AEs of any grade, such as veno-occlusive disease (VOD; 11% vs 1%), were more common in patients receiving InO vs chemotherapy.26
In a phase I/II study (NCT01371630), the addition of blinatumomab to InO + dose-reduced hyper-fractionated cyclophosphamide + vincristine + dexamethasone alternating with dose-reduced methotrexate + cytarabine chemotherapy in patients with R/R, Ph− B-ALL, gave an overall response rate of 80%, a CR rate of 57% and a best MRD negativity rate of 81%, similar to rates without blinatumomab.27 Common AEs included prolonged thrombocytopenia (76%), infection (53%), and nausea (35%). Hepatic AEs were reported in 33% of patients, including VOD in 10% of patients.27
Several studies investigating InO combination therapies in the first-line setting are underway, largely focusing on older adult patients.25 In the ongoing Alliance A041703 study (NCT03739814), InO followed by blinatumomab is being evaluated in patients aged ≥60 years with ND Ph− B-ALL, with results from Cohort 1 demonstrating an 85% composite CR rate after two cycles of InO and 97% following two cycles of blinatumomab.28 At 1 year, the event-free survival (EFS) rate was 85% and the OS rate was 75%.27 Long-term follow-up of this study is warranted to confirm durability of responses.25
Results from these studies indicate that InO, alone or in combination with chemotherapy or immunotherapies, is a viable treatment option for patients with CD22+ B-ALL across age groups.
CAR T-cell therapies
CAR T-cell products are genetically engineered to include fusion proteins that guide them towards molecular markers on tumor cell surfaces, enabling selective targeting of cancer cells (Figure 3).29 Brexucabtagene autoleucel (brexu-cel) and obecabtagene autoleucel (obe-cel) are CAR T-cell therapies approved by the FDA and EMA for the treatment of adult patients with R/R BCP-ALL (Table 1).11,12,15,16 Tisagenlecleucel is the only FDA- and EMA-approved CAR T-cell therapy for the treatment of pediatric patients with ALL (Table 1).13,14 Although only currently indicated for R/R disease, integration of CAR T-cell therapies into first-line treatment in concomitant or sequential regimens is of interest to potentially increase cure rates and reduce the need for chemotherapy.29
Figure 3. Mechanism of action of CAR T-cell therapy*
.webp)
In the phase I/II ZUMA-3 trial (NCT02614066), evaluating the efficacy and safety of brexu-cel in adult patients with R/R BCP-ALL, at 3 years, the median CR rate was 63%, duration of response (DoR) was 14.6 months, and OS was 25.6 months in the overall population and 38.9 months in responders.31 At 5 years, the OS rate was 40%, with a median OS of 25.6 months in the overall population and 53.5 months in responders.32 The most common Grade ≥3 AEs were anemia and pyrexia.31–33
In a retrospective study by the Real-World Outcomes Collaborative of CAR-T in Adult ALL (ROCCA) consortium, which was formed to study brexu-cel in patients with R/R B-ALL as a standard-of-care therapy, 90% of 168 efficacy-evaluable patients achieved a CR/CR with incomplete hematologic recovery (CRi), and 79% an MRD− CR.34 At a median follow-up of 11.4 months, 6- and 12-month progression-free survival (PFS) rates were 59% and 48%, respectively; CRS was reported in 84% of patients and immune effector cell-associated neurotoxicity syndrome (ICANS) in 56%.34
Results from the phase Ib/II FELIX trial (NCT04404660), evaluating obe-cel in adult patients with R/R BCP-ALL, showed that CR and CRi was achieved by 55% and 21% of patients with morphological disease, respectively.35 The median response duration in patients who received ≥1 obe-cel infusion was 21.2 months, and the median EFS was 11.9 months.35 Grade ≥3 AEs included CRS (2.4%) and ICANS (7.1%).35 In the follow-up analysis of patients who were studied for ≥3 years, 40% of responders were in ongoing remission without additional therapeutic intervention.36
A ROCCA analysis, investigating real-world utilization of obe-cel and brexu-cel in patients with R/R ALL, found that response rates were similar between patients treated with obe-cel vs brexu-cel (p = 0.85), with MRD− CR/CRi rates of 81% vs 80%, respectively.37 However, rates of CRS (56% vs 94%; p < 0.0001) and ICANS (17% vs 51%; p = 0.001) were lower in patients treated with obe-cel vs brexu-cel.37
These studies support CAR T-cell therapy as a therapeutic option for the treatment of patients with R/R B-ALL; however, further research into its use in other ALL lineages and younger patient populations is warranted to expand usage.
Immunotherapies in combination with TKIs
Combination therapies of TKIs with chemotherapy are routinely used in first-line regimens for Ph+ ALL,2 and are now being investigated in combination with immunotherapies.38 They act by blocking tyrosine kinase and other intracellular kinases in order to regulate cell growth and angiogenesis, preventing disease progression. However, those that inhibit multiple kinases, in particular, have an increased risk of toxicities, and management of these toxicities is crucial to prevent long-term sequelae.39
A study of the efficacy and safety of blinatumomab + a TKI vs chemotherapy + a TKI in patients with de novo or relapsed Ph+ B-ALL found that rates of OS, PFS, and non-relapse mortality (NRM) were similar between treatment groups (2-year OS, 87% vs 78%; PFS, 81% vs 54%; NRM, 6.3% vs 14%), although NRM was numerically higher with chemotherapy.38 Severe treatment-related AEs were more common in patients receiving chemotherapy vs blinatumomab, suggesting blinatumomab + TKIs could be an effective and tolerable chemotherapy-free approach to treating this patient population.38
Implementing reduced chemotherapy and chemotherapy-free regimens into clinical practice
Deciding which reduced chemotherapy or chemotherapy-free regimen is most appropriate for each patient requires multifactorial consideration, with aspects such as age, ALL subtype, and treatment availability playing key roles.1,4
Elderly patients
Therapy selection for older or frail patients can be challenging due to more frequent adverse prognostic features and comorbidities, and risks associated with treatment such as organ damage, immunosuppression, and exacerbation of underlying conditions.4 While blinatumomab has been shown to be effective in elderly patients and does not pose the same risk of myelosuppression as conventional chemotherapy, CAR T-cell products, or ADCs, further randomized comparative studies of blinatumomab vs chemotherapy and other combinations are warranted to confirm safety and tolerability.2,4,40 At present, blinatumomab with a TKI is considered to be a tolerable treatment option that provides durable remissions in elderly patients with ALL, eliminating the risk of chemotherapy-associated AEs.4 Furthermore, InO is efficacious and tolerable in older patients with ND B-ALL, with improved response and survival rates over chemotherapy, suggesting that integration into first-line regimens may be a viable for this population.41 In addition, brexu-cel may offer another chemotherapy-free option in this population.4
Pediatric patients
Pediatric patients with ALL are usually treated according to risk, identified through bone marrow or peripheral blood testing for genetic abnormalities, which determines the intensity of therapy required.42 While they are generally able to tolerate chemotherapy better than adult patients,3 reducing chemotherapy through the introduction of immunotherapies has been of great clinical interest given the potential to improve outcomes.43 Blinatumomab has shown efficacy and tolerability in pediatric patients with SR B-ALL,22 leading to its approval by the FDA and EMA for patients aged ≥1 month (Table 1).7,8 Similarly, InO has demonstrated encouraging efficacy and safety results in younger patients with Ph− B-ALL,25 and is approved by the FDA for pediatric patients aged ≥1 year with BCP-ALL (Table 1).9 While studies evaluating brexu-cel, obe-cel, and other CAR T-cell therapies in pediatric patients with ALL are ongoing,43 tisagenlecleucel is the only CAR T-cell therapy available in this population (Table 1).13,14
Ongoing unmet needs and future directions
Use of response-based factors, such as MRD, to guide ALL treatment have been of particular interest in recent years.44 Integration of immunotherapies into ALL treatment regimens may help address the challenge of remaining MRD following traditional chemotherapy.1 A meta-analysis, assessing the prognostic impact of MRD in ALL, found that 10-year DFS in patients who achieved MRD negativity was 64% vs 21% in those who were MRD+ after induction and/or consolidation therapy.44 Factors such as MRD assay sensitivity/specificity, timing of assessment, and disease-related features may affect the prognostic significance of assessment, warranting further studies into MRD-guided therapy.44
There are some access barriers to immunotherapies, such as lack of familiarity with guidelines, healthcare infrastructure, and socioeconomic background.45 These barriers are often amplified in specific populations such as racial/ethnic minorities and elderly patients, impacting outcomes and patient experience.45 However, outpatient administration of blinatumomab to pediatric patients has shown improvements in symptom burden and quality of life for both patients and caregivers,46 and outpatient administration of brexu-cel in patients with B-ALL and mantle cell lymphoma demonstrated similar efficacy to inpatient delivery, with no increase in NRM or toxicities.47 These studies suggest that at-home or outpatient delivery of immunotherapies to treat ALL is a feasible option and should be explored further to improve patient experience.
Conclusion
While chemotherapy remains central to ALL treatment,2 broad cytotoxicity, poor tolerability in older patients, and MRD persistence provide compelling rationale for reducing or eliminating its use.3,5 Integration of immunotherapies, such as BiTEs, ADCs, and CAR T-cell therapies, as monotherapies or in combination with chemotherapy or TKIs, offers a significant step towards improved outcomes for patients across age groups and disease subtypes.1 Continued investigation through clinical trials, long-term follow-up studies, and real-world evidence will be essential to refine treatment algorithms, address ongoing unmet needs, and ultimately deliver effective, tolerable therapies to patients with ALL.
This educational resource is independently supported by Amgen. All content was developed by SES in collaboration with an expert steering committee. Funders were allowed no influence.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content




