All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know ALL.
The ALL Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the ALL Hub cannot guarantee the accuracy of translated content. The ALL Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The ALL Hub is an independent medical education platform, sponsored by Amgen and Autolus. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View ALL content recommended for you
Targeted versus conventional therapy in R/R Ph-like ALL
Patients with Philadelphia (Ph) chromosome-like B-cell acute lymphoblastic leukemia (B-ALL) have a similar gene expression profile to those with Ph-positive ALL; however, they lack a BCR::ABL1 translocation.1 Treatment with conventional chemotherapies can result in poor outcomes due to CRLF2 overexpression in patients with Ph-like ALL; therefore, it is important to evaluate the potential of novel and targeted therapies in these patients.
The ALL Hub has previously reported on treatment strategies for Ph-like ALL. Here, we summarize a recently published article by Desikan et al.1 evaluating the outcomes of adult patients with relapsed/refractory CRLF2-rearranged B-ALL, published in American Journal of Hematology.1
Study design1
This retrospective analysis including patients with relapsed/refractory CRLF2-rearranged Ph-like ALL treated at the MD Anderson Cancer Center, Houston, US, between 2001 and 2021. Patients entered the study when they received their first therapy. CRLF2 expression, prior treatments, cytogenetics, and Janus kinase 2 (JAK2) mutational status were assessed for all patients. The study endpoints were relapse-free survival (RFS), calculated from response to salvage therapy to subsequent relapse or death, and overall survival (OS), calculated from study entry to death.
Results1
Overall, 69 patients were included in the analysis, of which 41 patients had a CRLF2 overexpression and 26 patients having an accompanying CRLF2 fluorescence in situ hybridization performed; all 26 of these patients had a CRLF2 rearrangement. Patients were split into two cohorts based on their first salvage therapy at the institution with or without chemotherapy (targeted therapy cohort), or conventional chemotherapy (conventional therapy cohort). Targeted therapies included inotuzumab ozogamicin and/or blinatumomab and/or CAR T-cell therapy. Patient characteristics are shown in Table 1. The majority of patients were male and Hispanic.
Efficacy
Median follow-up was 46 months. The rate of complete remission/complete remission with incomplete count recovery is shown in Figure 1, with overall survival (OS) shown in Figure 2.
Table 1. Baseline patient characteristics*
|
CAR, chimeric antigen receptor; JAK2, Janus kinase 2; SCT, stem cell transplant. |
||
|
Characteristic, % (unless |
Targeted therapy cohort |
Conventional therapy cohort |
|---|---|---|
|
Median age (range), years |
39 (18–69) |
35 (18–72) |
|
Male |
71 |
71 |
|
Hispanic |
61 |
76 |
|
Median prior lines of therapy (range), n |
1 (1–5) |
1 (1–4) |
|
Prior allo-SCT |
11 |
15 |
|
Prior inotuzumab/blinatumomab |
29 |
22 |
|
Prior CAR T-cell therapy† |
7 |
0 |
|
Disease status |
|
|
|
Primary refractory |
14 |
10 |
|
Salvage 1 |
64 |
71 |
|
Salvage ≥2 |
22 |
19 |
|
JAK2 mutated‡ |
22 |
50 |
Figure 1. Response rates*
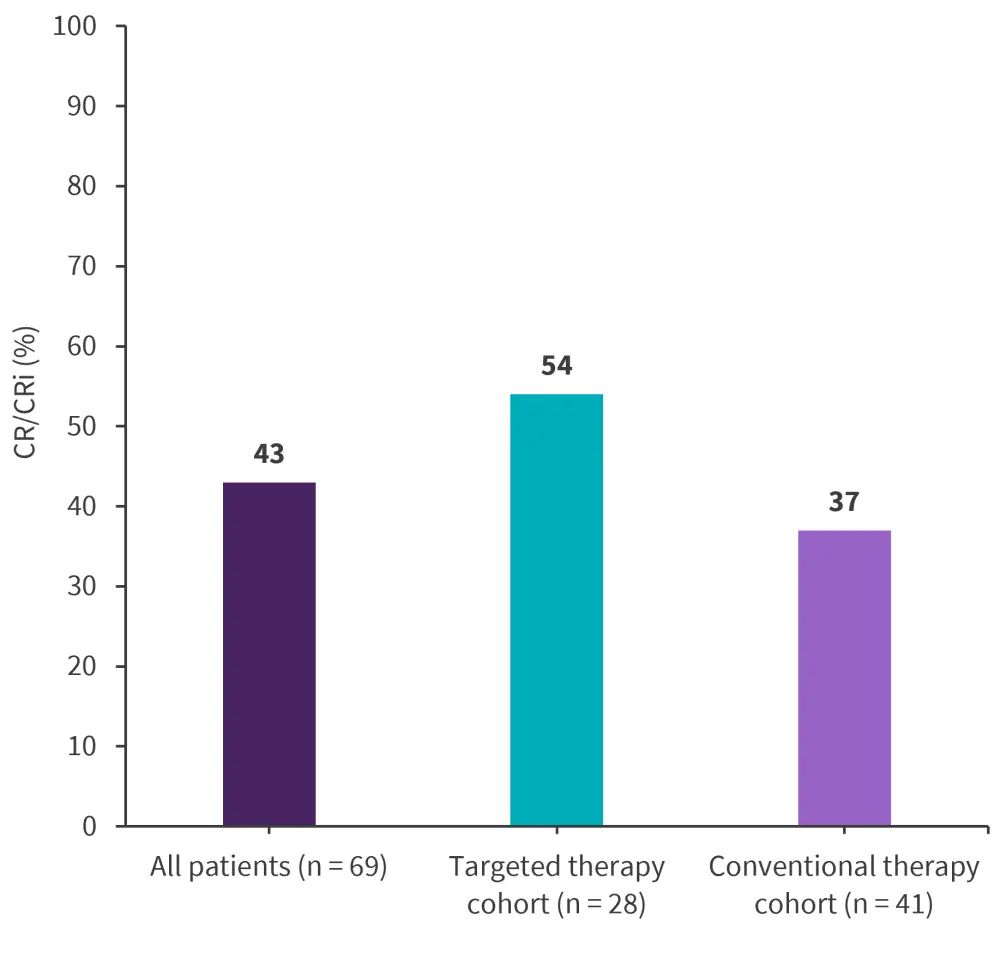
CR/CRi, complete remission/complete remission with incomplete count recovery.
*Adapted from Desikan, et al.1
Figure 2. Overall survivial*
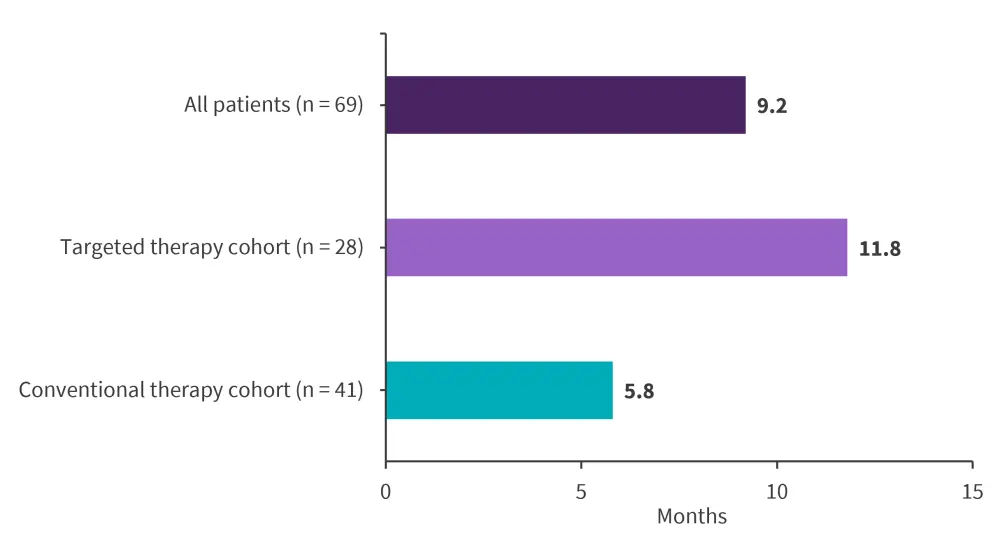
*Adapted from Desikan, et al.1
When OS was analyzed based on JAK2 mutation status, patients with JAK2 mutations had a shorter median OS compared to patients with wild-type mutations (5.8 vs 10.5 months, respectively).
Conclusion1
This study demonstrated that targeted therapies can improve outcomes in patients with Ph-like ALL, compared with patients treated with conventional chemotherapy. Although OS was higher in patients treated with target therapy, this difference was not statistically significant, potentially due to the small cohort sizes. Several limitations were noted, including patients entering the study at different time points during salvage therapy and patients receiving various prior therapies; this potentially restricted the evaluation of each therapy type and associated outcomes. Further, larger studies are warranted to evaluate the addition targeted therapies into Ph-like ALL treatment, providing insight into the optimal treatment regimen for these patients.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content


